Global-English



Craftsmanship and benevolence
We uphold the pure heart of protecting health and putting life first,
keenly understanding the pain points of the industry, and dedicating ourselves to tackling key technologies.

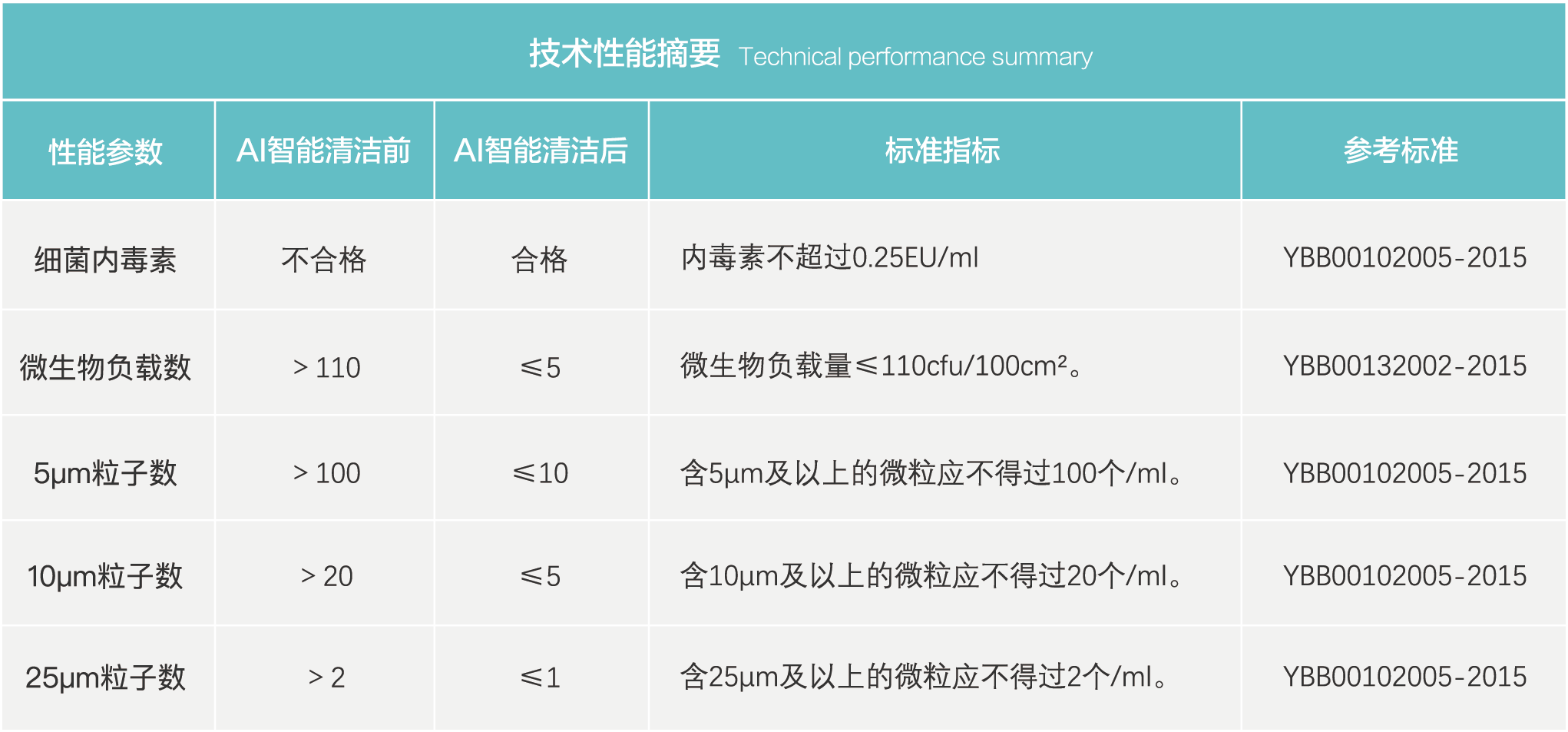
The main features are dry, low-temperature, fast, thorough, and environmentally friendly. At the same time, the elimination of static electricity and hydrophobicity on the surface of the material has been achieved, achieving hydrophilicity. In the production of pharmaceutical packaging materials, CEP "new ultra clean" technology has been used to 99.99% remove insoluble particles, bacteria, endotoxins and other hazardous substances on the material, making the medical process safer and more controllable.